Elmiron Linked to Retina Damage and Vision Loss
Elmiron, an oral medication prescribed for the treatment of interstitial cystitis, has been linked to permanent retinal damage and vision loss. At least 63 lawsuits have been filed against Johnson & Johnson subsidiary Janssen Pharmaceuticals, which manufactures Elmiron. Those lawsuits have been consolidated and transferred to one federal district in New Jersey. More lawsuits are expected to follow.
If you or your loved one has developed vision loss, blurred vision, pigmentary maculopathy, or any other severe injuries, contact Merkel & Cocke, P.A. today to learn more.
What is Elmiron prescribed for?
Elimiron (pentosan polysulfate sodium, or PPS) is prescribed to treat the symptoms associated with interstitial cystitis (IC), “a chronic condition causing bladder pressure, bladder pain and sometimes pelvic pain. The pain ranges from mild discomfort to severe pain. The condition is a part of a spectrum of diseases known as painful bladder syndrome.” The drug does not actually cure the condition, but it eases the symptoms by preventing substances in urine from irritating the lining of the bladder.
It is estimated that at least 1 million people have IC. Elmiron, which entered the market in 1996, is the only FDA approved medication to treat the symptoms of IC. There are no generic versions of the drug. This means that anyone who has IC and who is prescribed medication to treat the symptoms will be prescribed Elmiron.
What are the side effects of taking Elmiron?
Elmiron, like any prescription drug, has a host of potential side effects. They include:
- Nausea
- Diarrhea
- Headache
- Rash
- Dizziness
- Bruising
- Bloody stools
- Abnormal liver function
- Hair loss
Elmiron and pigmentary maculopathy
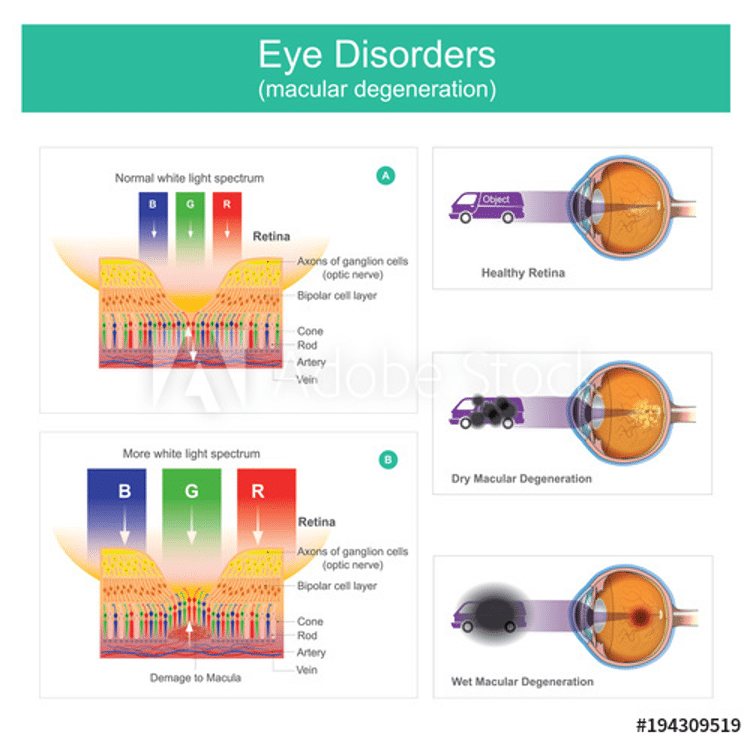
One of the most serious side effects linked to Elmiron is vision loss, caused by pigmentary maculopathy, a type of macular degeneration associated solely with PPS. The macula located in the retina, and “is the functional center of the retina. It gives us the ability to see ‘20/20’ and provides the best color vision.” Degeneration of the macula leads to vision loss. Sometimes this loss starts of the periphery and moves inwards; often, it starts in the center of the eye and moves outwards, creating a halo-like effect, where only the peripheral vision remains.
Per research from the American Academy of Ophthalmology (AAO), “about a quarter of patients with significant exposure to the drug [Elmiron] show signs of retinal damage.”
Symptoms of vision loss associated with Elmiron
Patient who took Elmiron have reported:
- Blurry vision
- Dimmed vision
- Difficulty adjusting to changes in brightness and light
- Vision loss in the center of their field of vision
- Difficulty reading
- Nearsightedness
It took Janssen two years to warn patients about the risk of vision loss
The AAO research was published in 2018, but Janssen did not start issuing warnings about the risks until June 2020. This means that potentially thousands of people have sustained permanent vision loss after taking Elmiron over the past 25 years.
Can the vision loss be treated?
It depends on how severe it is. The AAO found that patients who were just showing signs of retinal damage from drug toxicity could potentially mitigate the damage. Some patients may still have difficulty with blurry vision or reading small print, but they will not lose their sight. However, macular degeneration is an irreversible condition even when it is not brought on by a dangerous drug, and “In the late-stage, toxicity can mimic late-stage dry atrophic age-related macular degeneration and result in permanent vision loss.”
If I took Elmiron, can I file a lawsuit?
If you took Elmiron and suffered injury and losses associated with the drug, then you can file a product liability lawsuit. A federal judge has blocked a class action claim for patients who have not suffered harm, but who must undergo medical monitoring because of Elmiron use. At this time, you must have suffered an actual injury in order to proceed with a lawsuit.
As of January 2021, at least 80 lawsuits have been filed – but potentially thousands more may be coming. Many of those lawsuits have been consolidated into the District of New Jersey and will be heard by Judge Brian Martinotti.
Why choose Merkel & Cocke, PA for an Elmiron lawsuit?
Since 1982, Merkel & Cocke, P.A. has earned a reputation for handling complex litigation that other firms may not be able to take on. We have the experience, resources, and skill to handle class action, MDL and individual lawsuits for dangerous drugs and defective medical devices. We have secured millions of dollars on behalf of our product liability clients. Our experienced team of litigators will fight aggressively and proactively on your behalf.
If you took Elmiron and have sustained vision damage or vision loss, or been diagnosed with retinal damage caused by drug toxicity, Merkel & Cocke, P.A. is ready to fight for you. You don’t have to face off against the drug companies alone. Our Mississippi product liability lawyers are here to assert your right to justice and to compensation for your injuries. To schedule a free consultation about your case, please call us at 662-627-9641, or fill out our contact form. From our offices in Jackson, Clarksdale, Greenville, and Oxford, we are proud to represent clients throughout Mississippi and the Gulf Coast Region. We also handle cases in Tennessee and Arkansas, and in federal courts in all three states.

After graduating from the University of Virginia Law School in 1975, Mr. Cocke and Mr. Merkel established Merkel & Cocke, P.A. in 1982. Since that time the emphasis of Mr. Cocke’s practice has progressed toward medical malpractice. At the present time his practice is exclusively devoted to handling medical negligence cases for the plaintiff, either as a result of direct contact by the client or on referral from other attorneys who are not familiar with the handling of medical negligence cases. Mr. Cocke was selected Best Lawyer of the year for 2012 and 2014 in The Memphis area in the field of Medical Malpractice and has been selected a Best Lawyer and Super Lawyer every year since 2006. Learn more about John Hartwell Cocke here.